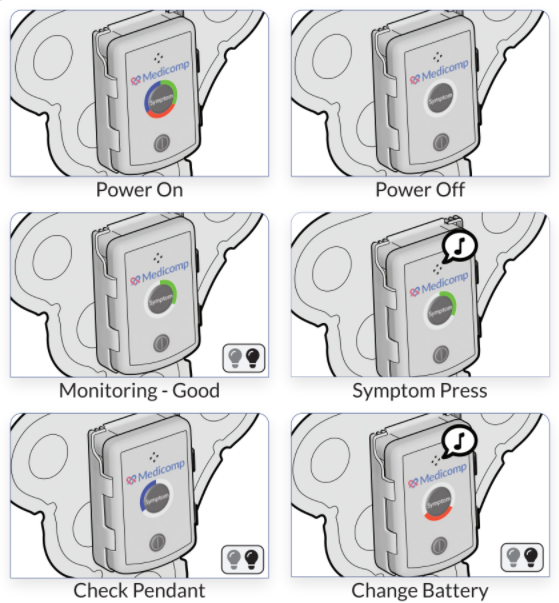
Though the amount of time between a medication’s initial publication and its actual release may seem endless, the need to properly test the medication, service, or treatment using humans rather than mice or other vertebrates is essential. ReactDx has a long-standing history with organizations as they initiate ECG clinical trials to determine whether their newest product or service fills a need without inadvertently harming patients.
Before clinical trials begin, medicines and treatments must be created, modified, and tested in preclinical trials, which generally involve non-human subjects. For every 1,000 potential medicines tested in preclinical trials, only one moves on to a clinical trial. Most medicines are tested for 6 years or more before a clinical trial begins. Clinical trials are generally a group effort involving design ideas from doctors, scientists, other health care professionals, and patients. Although most people view a clinical trial as how a specific medication affects different individuals, clinical trials may focus on new surgical procedures, physiotherapy, rehabilitation, chemotherapy, complementary therapies, behavioral therapy, et cetera.
Clinical trials generally involve 3 main phases. During Phase I, a scant number of patients are invited to trial a new medication to determine its effect. Researchers judge how safe the drug is and whether it should continue to the next phase. Participants may be either healthy individuals or ill patients who have explored all other options. The medication or treatment is given to all participants.
Phase II picks up the pace with a larger pool of patients to focus on safety and side effects and to determine whether the medication delivers positive results. By Phase II, the new medication or service may be compared to another well-established treatment.
Phase III is much larger, encompassing a vast number of patients to measure the results of the new method or medication to the old and determining which is the better prospect. Any side effects are more easily assessed with a larger pool of patients. Phase III trials are usually randomized clinical trials involving at least one experimental group and one control group. The accuracy and reliability of a clinical trial is solidified with a blind trial, in which the patient is not told whether he or she is receiving a new drug or treatment or a placebo. In a double-blind trial, neither the doctor nor the patient is aware if the medication is a placebo or the trial’s medication.
ReactDx’s clinical research department works with various entities as they go through the ECG clinical trials process. With over a dozen available services, ReactDx can give your clinical trial the support and security it needs with a measurable analysis of patient heart stress. Contact ReactDx at 800-23-HEART and read our blog to learn about our past involvement in clinical trials, and how we can assist your organization in its quest to improve patient health.