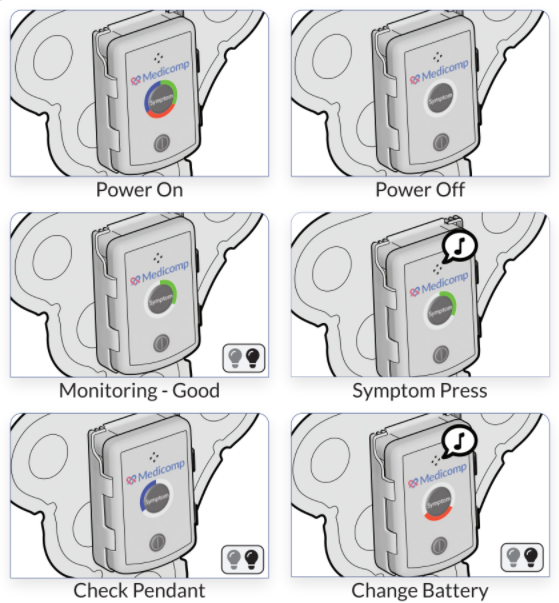
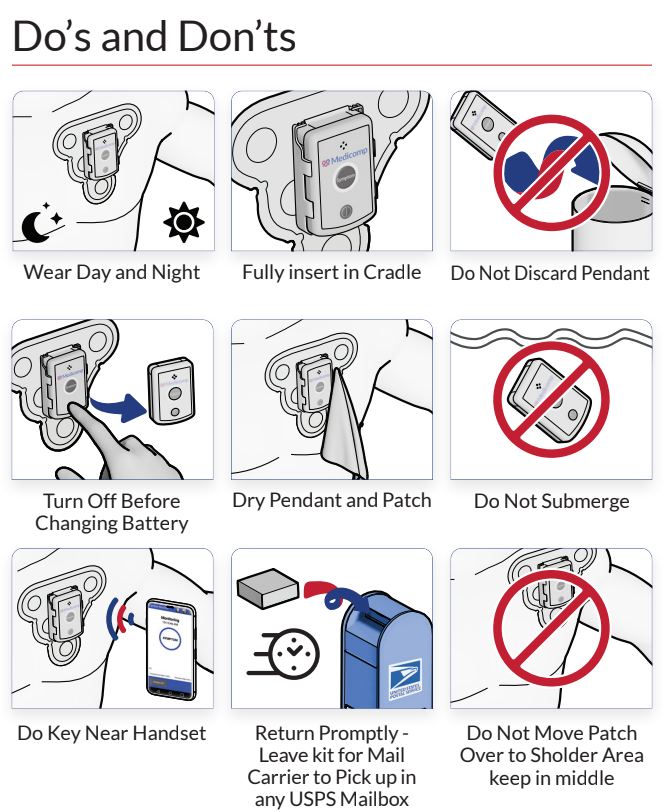
The Mayo Clinic reports that stem cell therapy works by growing healthy stem cells in a lab and then manipulating them to match specific cells within the body, such as heart muscle cells. These cells are then injected into an ill patient so that they can begin healing and repairing the targeted problem.
The Old FDA Vs. the New FDA
The old FDA guidelines were very strict when it came to stem cell therapies. Researchers were required to conduct clinical trials that proved the therapy was safe and effective. Since then, the 21st Century Cures Act was passed, which allows cell therapies to be used to treat individuals with severe or life-threatening illnesses. Additionally, President Trump appointed Scott Gottlieb as the new head of the FDA. Gottlieb is known to be more lenient than previous FDA commissioners and has expressed his willingness to lower the guidelines for approving such therapies.
Mesoblast
The first company that is ready to take advantage of the new FDA guidelines is Mesoblast. The company will conduct two clinical trials to aid individuals who have been placed in either the Class III or Class IV stage of heart failure. The first trial will involve 159 patients with end-stage heart failure, while the second trial studies 600 participants mild to moderate chronic heart failure. Both trials will be completed by mid-2018.
Capricor
Another company that plans to move forward thanks to the lower guidelines is Capricor. Founded by Cedars-Sinai researcher Eduardo Marbán, Capricor seeks to treat hypertrophy that is associated with Duchenne muscular dystrophy. With Duchenne muscular dystrophy, muscles die off because they lack the protein dystrophin. The HOPE Trial will take a full year to complete.
Doctors would do well to stay informed in regards to the new stem cell therapy trials. If these therapies are proven effective, it may not be long before access to the treatment becomes more accessible. ReactDx’s cutting-edge Holter monitoring equipment is also an important tool that aids cardiologists in getting a better picture of a patient’s heart health. Visit ReactDx’s News & Updates page for the latest information, or give us a call at 800-234-3278.
At ReactDx, we have been advancing diagnostic cardiology since 1981. ReactDx develops, manufactures, and provides service with the most reliable and sophisticated Holter monitoring systems in the world. Since its inception, ReactDx has been at the forefront of the cardiac telemedicine industry – pioneering technology, improving existing technologies and adding new applications.