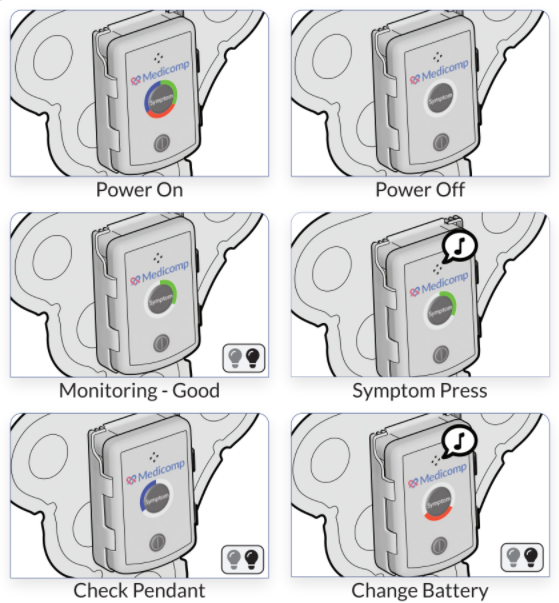
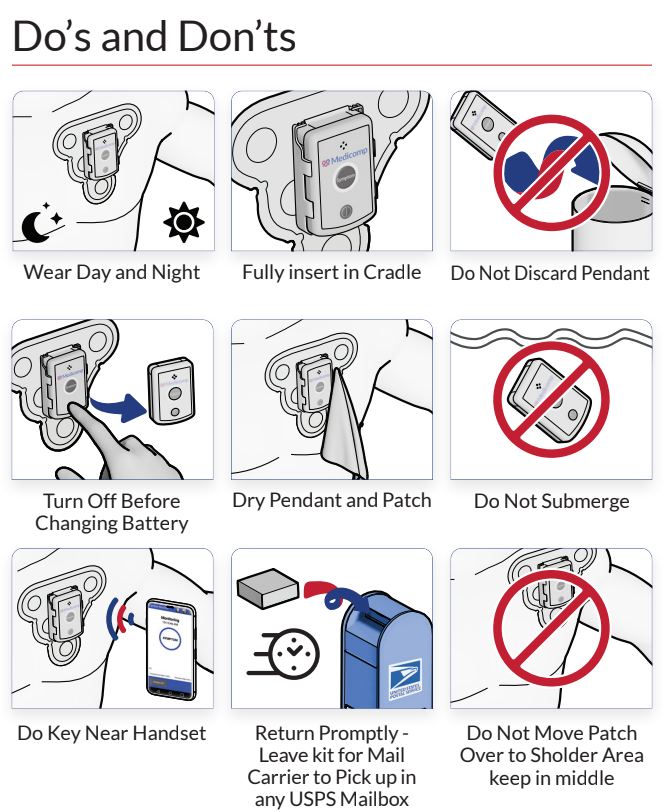
About ReactDx
Welcome to ReactDx (React Diagnostics), a leading innovator in the cardiac telemedicine and home sleep testing industries. As a trailblazer in both fields, we take pride in being developers, manufacturers, and service providers at the forefront of cutting-edge healthcare technology.
The ReactDx and React Health family understands the importance of accurate diagnostics and timely interventions. Patients deserve access to care they need without unnecessary delays to achieve a better quality of life as soon as possible.
ReactDx includes both Watermark Medical, Inc. and BioSerenity USA, Inc. Under the umbrella of ReactDx, these entities operate harmoniously to redefine the standards of remote cardiac and sleep disorder diagnostics. Our collective expertise and dedication ensure that patients receive the most accurate assessments from the comfort of their own homes.
Join us in our mission to revolutionize healthcare through innovation. With ReactDx, the future of ambulatory cardiac monitoring and home sleep testing is here.
Our Purpose
Our Purpose
ReactDx’s purpose is to help physicians add quality years to patients’ lives.
ReactDx’s purpose is to help physicians add quality years to patients’ lives.
Core Values
Core Values
- Passion for Our Purpose
- Integrity in All We Do
- Respect the Individual
- Intense Customer Focus
- Undeniable Quality
- Passion for Our Purpose
- Integrity in All We Do
- Respect the Individual
- Intense Customer Focus
- Undeniable Quality
Learn more about our Products & Services
React Diagnostics develops and produces its own products and systems using diagnostic technologies that are widely regarded as the most advanced in the industry. Our patented Diogenes algorithm is unmatched in discovering asymptomatic arrhythmias, providing beat-to-beat analysis of the P-wave as well as Rate, Rhythm, and QRS morphology.